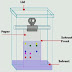
Chromatography is a
modern separation procedure that consists of different physical techniques and
used in many industrial sector especially in the chemical and pharmaceutical sector.
This process is mainly applied for the separation of organic compounds mixture.
Though, a number of other separation processes are there, applied in the
purification process but when it comes to separating two similar physical and
chemical nature organic compounds,...